Research Westermann Group: The Molecular Machinery of Mitochondrial Dynamics, Inheritance, and Architecture
Each type of eukaryotic cell has a characteristic three-dimensional structure. Maintenance of its architecture and duplication during cell division depend on active transport of organelles along the cytoskeleton and continuous fission and fusion of intracellular membranes. Mitochondria are essential organelles, which are often located at sites of high energy consumption in the cell. They cannot be formed de novo, but have to be inherited during cell division. Positioning and transport of mitochondria and maintenance of mitochondrial function are controlled by the cytoskeleton and depend on fusion and fission of their membranes. These processes are functionally linked to the machinery that maintains the complex architecture of mitochondrial inner membrane cristae. We use the budding yeast Saccharomyces cerevisiae to study these processes. By studying mitochondrial morphology and inheritance we can learn a lot about many different aspects of organellar biogenesis, such as partitioning of organelles during cell division, transport of membranes along the cytoskeleton, intracellular membrane fusion, cellular aging and much more.
Asymmetric inheritance of mitochondria in yeast (DFG We 2174/7-1)
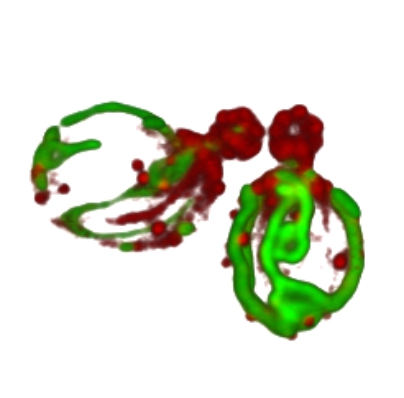
Mitochondria are essential organelles of eukaryotic cells. They have their own genome, which encodes a handful of mitochondrial proteins, and cannot be generated de novo. The inheritance of mitochondria during cell division requires the choreography of membrane fission and fusion processes with cytoskeleton-dependent transport of the organelles. In some cell types, mitochondria are partitioned asymmetrically, i.e. the daughter cells receive organelles with different properties. This serves, for example, to pass on healthy and fit mitochondria to the daughter cell that is determined to undergo a larger number of cell division cycles. Also yeast cells have the remarkable capacity to retain old and damaged mitochondria in the mother cell, while at the same time fit and metabolically active mitochondria are transported to the bud. The molecular mechanisms that determine the asymmetric distribution of mitochondria and the selection of healthy organelles for inheritance are largely unknown and are being studied using the yeast Saccharomyces cerevisiae as a model system. In our current work, we have developed an assay that allows us to observe the inheritance and dynamics of individual oxidatively damaged mitochondria in vivo. We were able to show that the adaptor protein Mmr1 acts as a molecular switch that regulates the binding of the myosin motor protein Myo2 to mitochondria. By releasing Myo2/Mmr1, damaged mitochondria are uncoupled from the actin-dependent transport machinery and thus immobilized in the mother cell. We have identified Alo1 as another mitochondrial outer membrane protein that is particularly important for mitochondrial inheritance during oxidative stress. We would now like to decipher the signals and mechanisms that recognize the oxidative damage in mitochondria and trigger the immobilization of dysfunctional organelles. We will then investigate the significance for cellular ageing. Furthermore, we will focus on the consequences of damage in the mitochondrial DNA as well as the role of cell cortex anchors and retrograde transport of mitochondria. This work is expected to provide a detailed understanding of the processes that determine the asymmetric inheritance of mitochondria and thus ageing of the cell.
Sequence Information for mtGFP Plasmids
